 by Natalie Z. Sun, MD; Teresa Ro, BS; Puneet Jolly, MD, PhD; Christopher J. Sayed, MD
by Natalie Z. Sun, MD; Teresa Ro, BS; Puneet Jolly, MD, PhD; Christopher J. Sayed, MD
All authors are with the University of North Carolina at Chapel Hill, Dermatology, in Chapel Hill, North Carolina.
Funding: No funding was provided for this study.
Disclosures: The authors have no conflicts of interest relevant to the content of this article.
Keywords: Pyoderma gangrenosum; hidradenitis suppurativa; acne inversa; PASH; anti-IL-1 therapy, anti-TNF therapy, canakinumab
Abstract: Pyoderma gangrenosum and hidradenitis suppurativa are skin conditions characterized by an intense neutrophil-mediated inflammatory response that is often difficult to effectively treat. Successful use of interleukin (IL)-1beta inhibition using canakinumab and anakinra has been reported in patients with concomitant pyoderma gangrenosum and hidradenitis suppurativa. We report two cases where targeted therapy with canakinumab failed to lead to improvement for patients with pyoderma gangrenosum and hidradenitis suppurativa. The reason behind the non-response to IL-1beta blockade seen in these patients is unclear. Our report suggests that further controlled studies are warranted to help clinicians predict treatment responses to anti-IL-1 therapies in these challenging patients.
J Clin Aesthet Dermatol. 2017;10(9):36–38
Introduction
Pyoderma gangenosum (PG) and hidradenitis suppurativa (HS) are inflammatory skin conditions that sometimes co-exist along a spectrum of autoinflammatory syndromes.1 Positive responses to blockade of interleukin (IL)-1beta activity using canakinumab and anakinra have previously been reported in patients with concomitant PG and HS.[1, 5–8] We report two patient cases with difficult-to-treat PG and HS where targeted therapy with canakinumab, a monoclonal anti-IL-1beta antibody, failed to lead to clinical improvement.
Clinical Presentation
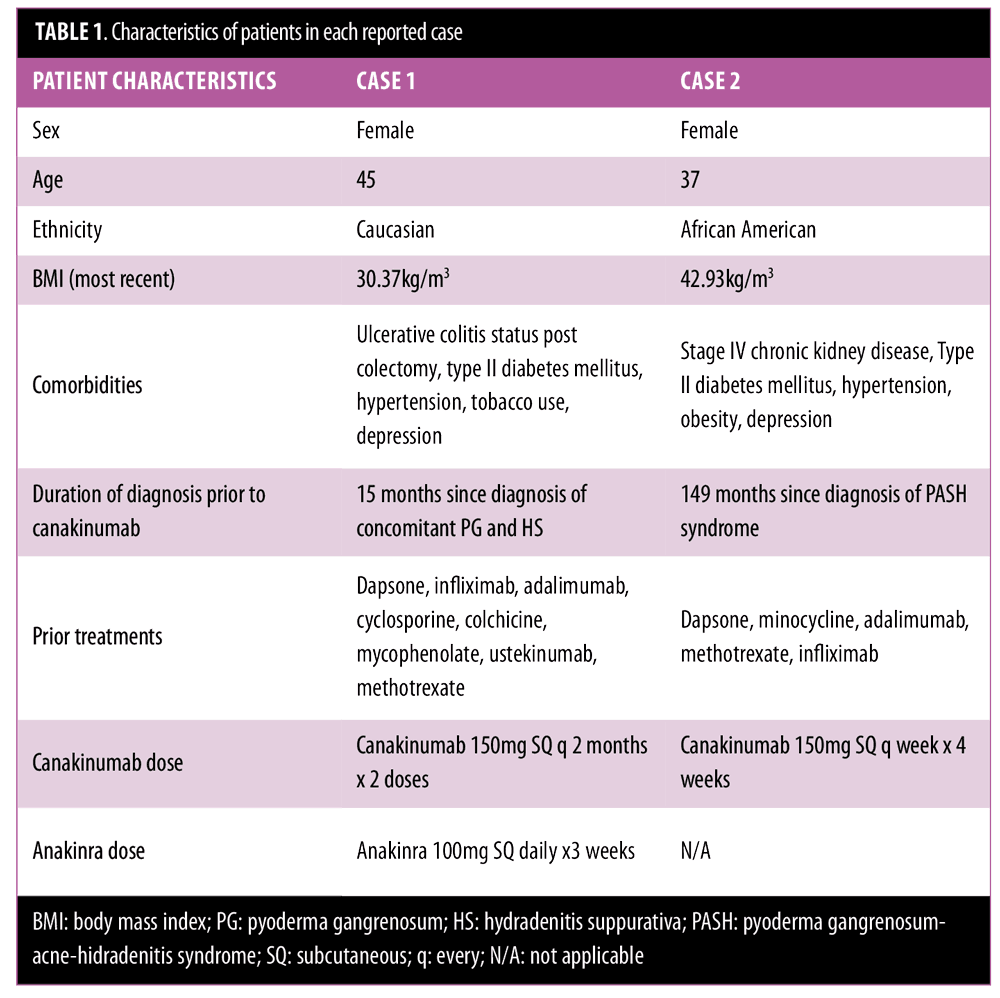
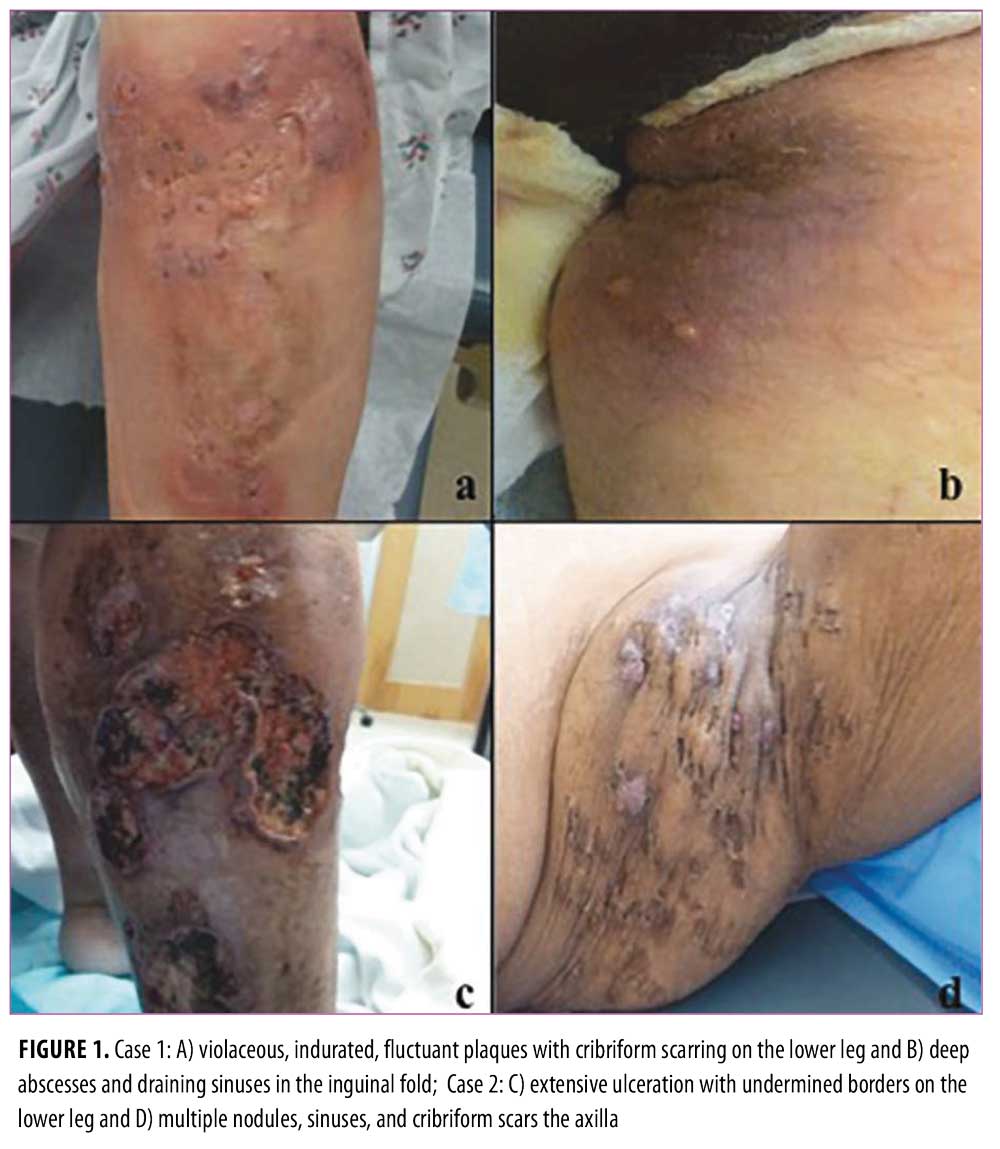
Case 1. A 45-year-old woman with PG of her left lower leg and HS in her bilateral groin presented frequently to our clinic for recalcitrant disease. Initially she responded well to infliximab 5mg/kg intravenously (IV) at Weeks 0, 2, and 6 of treatment, but then developed a hypersensitivity reaction with the third dose. Infliximab was discontinued, after which she demonstrated intolerance or failure to respond to several systemic treatments (Table 1).
Canakinumab was initiated at 150mg subcutaneously every eight weeks. Treatment was discontinued after two injections due to loss of insurance and lack of response. During the treatment period, the patient was admitted to the hospital twice for exacerbation of her PG. She also did not improve with anakinra 100mg daily for three weeks, which was administered 25 months after the treatment attempt with canakinumab.
Case 2. A 37-year-old woman presented with more than a 10-year history of PG affecting her left dorsal foot, bilateral lower extremities, and lower abdomen; HS in her bilateral axilla and left buttock; and acne, which is consistent with pyoderma gangrenosum-acne-hidradenitis (PASH) syndrome. She was being administered long-term prednisone therapy, but failed to improve with many treatments (Table 1). She partially improved with adalimumab 40mg weekly, and then had complete healing of her PG with infliximab 6mg/kg IV every four weeks. Due to suspicion for drug-induced granulomatous interstitial nephritis, infliximab was discontinued with rapid return of symptoms. Subcutaneous administration of canakinumab 150mg weekly was initiated for four weeks, but her PG severely worsened during treatment. She quickly responded to re-introduction of infliximab without worsening kidney function in the following months.
Discussion
PG is an uncommon inflammatory neutrophilic disorder that presents as a painful, chronic, ulcerating skin lesions. It most typically presents on the lower limbs, but can appear anywhere on the body. It typically occurs in areas of minor trauma, and evolves rapidly from sterile papulopustules to painful necrotic ulcers.[4] PG is commonly associated with inflammatory bowel disease, rheumatism, and hematological diseases.[3] Its pathogenesis is not completely understood; however, insufficient pathways to protect the epidermis from neutrophil infiltration have been implicated as a possible etiology.[3] Histopathology of PG is overall nonspecific and varies on disease time course, although initial lesions show suppurative folliculitis with neutrophilic infiltrate.[3] Diagnosis is based on clinical presentation, histopathology, and exclusion of other diseases.[2]
HS, also known as acne inversa, is a chronic inflammatory disease of hair follicles that manifests as tender subcutaneous nodules, typically in the axillae, perineum, and inframammary regions.[4] HS is commonly associated with acne conglobata, acne vulgaris, Crohn’s disease, dissecting cellulitis of the scalp, obesity, pilonidal disease, and smoking.[4] perilesional skin of HS and implicated in its pathogenesis.[4] Histopathology of HS lesions is nonspecific. Diagnosis is made based on clinical presentation.[5]
Pyoderma gangrenosum and hidradenitis suppurativa individually are conditions that are challenging to treat. Both conditions are characterized by an intense neutrophil-mediated inflammatory response.[1] Cytokine profiles in patients with HS have shown elevated IL-1beta and tumor necrosis factor (TNF), making anti-TNF and anti-IL-1 therapies logical treatment options.[6] In rare cases, pyoderma gangrenosum and hidradenitis suppurativa occur concomitantly, posing a similar treatment challenge for clinicians.[5] PG and HS is also seen together in PASH syndrome, a hereditary autoinflammatory syndrome first characterized in 2012 by Braun-Falco ,et al.[1] One of the two patients reported in the study achieved partial remission of inflammatory activity with the IL-1 receptor anakinra, leading to the hypothesis that IL-1beta might be involved in the pathogenesis of PASH syndrome.[1,5] Another case series by Hsiao et al[6] demonstrated a 75-percent response of PG ulcers in a patient with both PG and HS within three weeks of initiating treatment with anakinra. A case report by Jaeger et al[7] demonstrated complete remission of refractory PG in a patient with both PG and HS following four doses of humanized monoclonal antibody against IL-1beta, canakinumab 150mg every 3 to 6 weeks.
Contrary to cases in the literature that have reported successful use of IL-1beta blockade in patients with PG and HS, our two patients did not respond to canakinumab.[1, 5–8] Moreover, our first patient had two hospital admissions while on canakinumab for PG lesions despite receiving more frequent dosing of canakinumab than is typically reported. The reason for the lack of response to IL-1 inhibition seen in our patients is unclear. These patients are unlikely to have developed neutralizing antibodies during their brief treatment courses. Patients with specific mutations in tumor necrosis factor (TNF)receptor-associated periodic syndrome (TRAPS) have been noted to be refractory to IL-1beta antagonism, but there have been no similar mutations among HS or PG patients reported thus far that would indicate resistance to treatment.[9]
It is worth mentioning that both of our patients demonstrated relatively greater improvement on TNF blockade with infliximab than on canakinumab. Prior studies have shown anti-TNF therapy to be an effective treatment option for patients with PG and/or HS.[10,11] An enhanced understanding of the pathogenesis of PG and HS will allow for better treatment selection for these patients. Additionally, why some respond to anti-IL therapies while others respond to anti-TNF agents are unclear. Further controlled studies and basic science research are needed to help clinicians predict treatment responses to anti-IL-1 and anti-TNF therapies for these challenging patients.
References
- Braun-Falco M, Kovnerystyy O, Lohse P, Ruzicka T. Pyoderma gangrenosum, acne, and suppurative hidradenitis (PASH)—a new autoinflammatory syndrome distinct from PAPA syndrome. J Am Acad Dermatol. 2012;66(3):409–415.
- Powell FC, Schroeter AL, Su WP, et al. Pyoderma gangrenosum: a review of 86 patients. Q J Med. 1985:55(217):173–186.
- Wollina U. Pyoderma gangrenosum- a review. Orphanet J Rare Dis. 2007; 2:19.
- Alikhan A, Lynch PJ, Eisen DB. Hidradenitis suppurativa: a comprehensive review. J Am Acad Dermatol. 2009; 60(4); 539–561.
- Zarchi K, Dufour DN, Jemec GBE. Successful Treatment of severe hidradenitis suppurativa with anakinra. JAMA Dermatology. 2013;149(10): 1192–1194.
- Hsiao JL, Antaya RJ, Berger T, et al. Hidradenitis suppurativa and concomitant pyoderma gangrenosum: a case series and literature review. Arch Dermatol. 2010;146(11):1265–1270.
- Jaeger T, Andres C, Grosber M, et al. Pyoderma gangrenosum and concomitant hidradenitis suppurativa – rapid response to canakinumab (anti-IL-1b). Eur J Dermatol. 2013; 23:408–410.
- Kolios AGA, Maul JT, Meier B et al. Canakinumab in adults with steroid-refractory pyoderma gangrenosum. Br J Dermatol. 2015;173:112.
- Cantarini L, Lopalco G, Vitale A, et al. Delights and let-downs in the management of tumor necrosis factor receptor-associated periodic syndrome: the canakinumab experience in a patient with a high-penetrance T50M TNFRSF1A variant. Int J Rheum Dis. 2015;18(4):473–475.
- Brooklyn TN, Dunnill MG, Shetty A, et al. Infliximab for the treatment of pyoderma gangrenosum: a randomized, double blinded, placebo-controlled trial. Gut. 2006;55(4):505–509.
- Martinez F, Nos P, Benlloch S, et al. Hidradenitis suppurativa and Chron’s disease: response to treatment with infliximab. Inflamm Bowel Dis. 2001;7(4):323–326.